
HUMAN CENTRED RESEARCH LTD
USER RESEARCH & HUMAN FACTORS CONSULTANTS
OUR PURPOSE
Human Centred Research’s (HCR) purpose is to support our clients to develop desirable, intuitive and safe products.
We do this by putting the human at the centre of the Design process.
We work collaboratively with consultancies, medical device manufacturers and pharmaceutical companies.
Providing support with all end-to-end user research and human factors activities.

ABOUT: OUR STORY

Who we are: Annmarie Nicolson is a User Research and Human Factors (HF) specialist with 10 years industry experience developing consumer products and medical devices.
Why we started: Human Centred Research Ltd (HCR) was created to support manufacturers deliver desirable, intuitive & safe solutions to market.
How we will do it: The vision of HCR is driven by 3 core values:
1. Work smart: Provide support to benefit our customer's business needs and end user needs simultaneously.
2. Work together: Collaborate with our customer's Human Factors and Design teams to deliver human centred research activities.
3. Champion the user: Continue to advocate Human Centred Design to drive the development of desirable, intuitive and safe products and medical devices.
Where: Currently based in Bristol, UK, working in a remote office space. Will travel to meet and collaborate with customers and to conduct user research.
SERVICES
Experienced applying a wide range of User Research and Human Factors methodologies throughout the design process, from exploration to validation.
Working to ANSI / AAMI / IEC 62366, ISO 14971, IEC 13485, MHRA & FDA HF Guidance
EXPERIENCE
Research: executed research from contextual inquiry to summative usability testing, across various user groups and user interfaces including hardware, software, connected devices, labelling and packaging.
HF Process: implemented Human Factors (HF) and Usability Engineering (UE) processes into the overall design development process, including writing HF standard operating procedures and documentation templates.
Use Related Risk: conducted Use Related Risk Assessments using a variety of tools and methods including uFMEA, HTA, Known Problems Analysis & more.
HF Documentation: experienced writing documentation from research planning to HF summary reporting. Can support with Use Specifications, UI Evaluation Plans, UI Specifications, User Requirements, Screeners, Consent Forms, Test Protocols, Discussion Guides, Formative / Validation Report, HF and UE Summary Reports.
HF Methods & Tools: used a vast range of user research methods and tools including: Contextual Inquiry, Attribute Mapping, Card Sorting, User Journey Mapping, Task Analysis, Expert Reviews, Cognitive Walkthroughs, Usability Testing & many more.
Regulatory: worked to US, UK & EU standards and guidance: ANSI / AAMI / IEC 62366, ISO 14971, IEC 13485, MHRA & FDA HF Guidance.
User & Device Categories: conducted user research with vulnerable patients, lay users, physicians, GPs, surgeons, nurses, adolescents, adults, geriatrics across categories including:
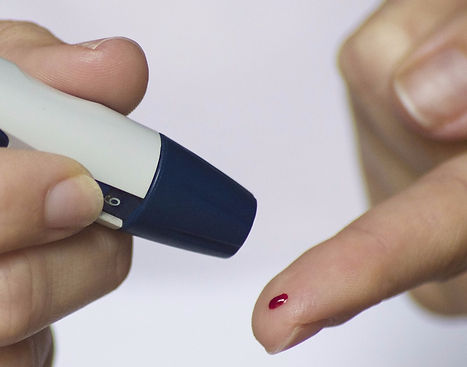
DIABETES MANAGEMENT
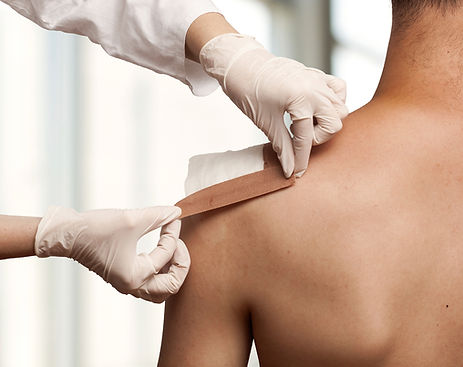
WOUND CARE

INHALATION DEVICES
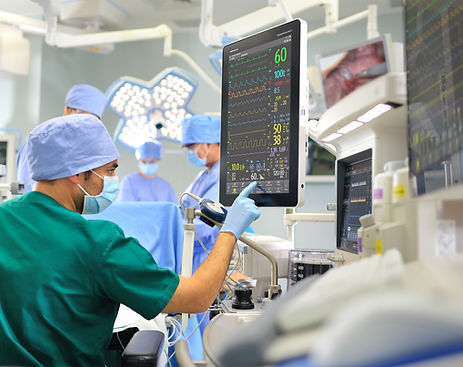
SURGICAL SYSTEMS
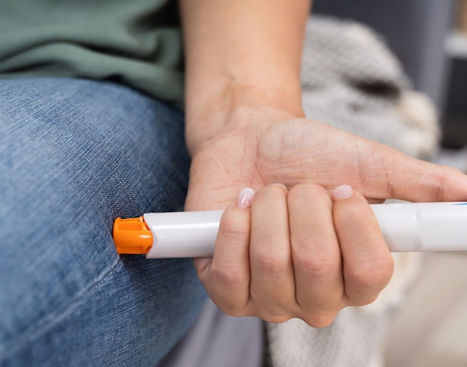
AUTO INJECTORS
